American academic freedom is in perilwww.science.org Academics researching online misinformation in the US are learning a hard lesson: Academic freedom cannot be taken for granted. They face a concerted effort—including by members of Congress—to undermi...
David A. Delgadillo
Organic-Natural Products-Medicinal-Analytical- Chemist. Postdoc in the Nelson Group @Caltech leveraging electron diffraction to discover novel natural products.
Discovery and Characterization of Pyridoxal 5′-Phosphate-Dependent Cycloleucine Synthasespubs.acs.org Pyridoxal 5′-phosphate (PLP)-dependent enzymes are the most versatile biocatalysts for synthesizing nonproteinogenic amino acids. α,α-Disubstituted quaternary amino acids, such as 1-aminocyclopentane-...
Synthetic biology identifies the minimal gene set required for paclitaxel biosynthesis in a plant ch...www.cell.com Genes specific for several steps of paclitaxel biosynthesis have not been identified,
hindering metabolic engineering of the full pathway in heterologous systems. This
study identifies four new genes ...
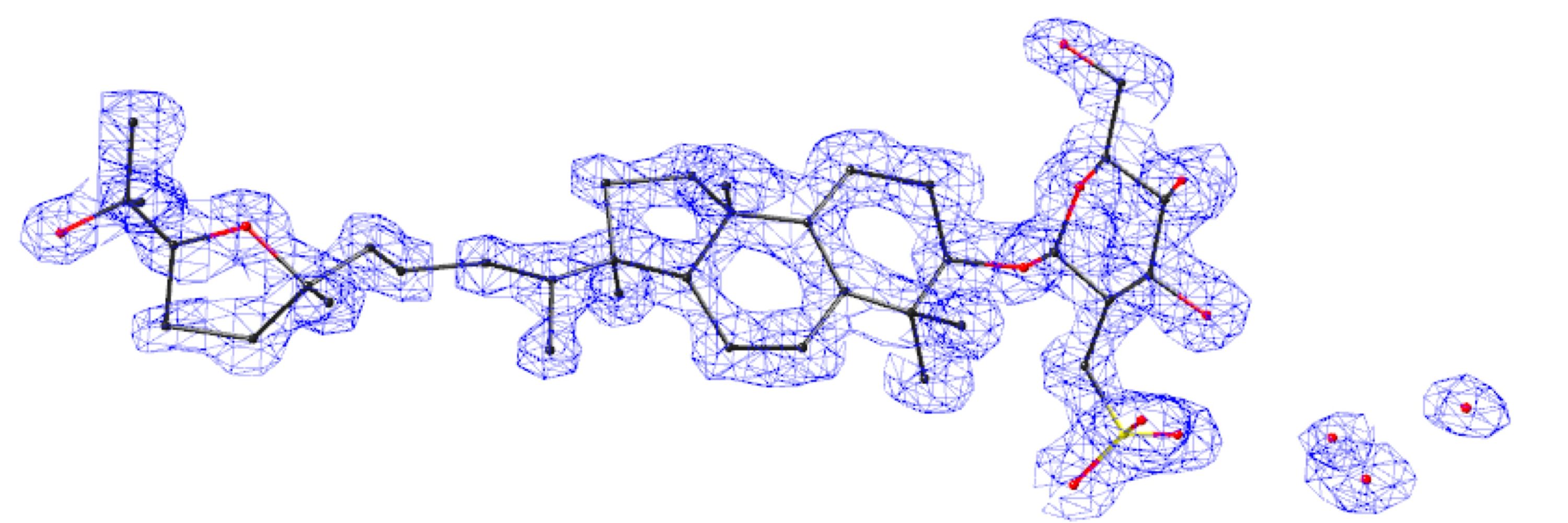
Biomimetic Synthesis and Chemical Proteomics Reveal the Mechanism of Action and Functional Targets of Phloroglucinol Meroterpenoidsdoi.org Natural products perennially serve as prolific sources of drug leads and chemical probes, fueling the development of numerous therapeutics. Despite their scarcity, natural products that modulate protein function through covalent interactions with lysine residues hold immense potential to unlock new therapeutic interventions and advance our understanding of the biological processes governed by these modifications. Phloroglucinol meroterpenoids constitute one of the most expansive classes of natural products, displaying a plethora of biological activities. However, their mechanism of action and cellular targets have, until now, remained elusive. In this study, we detail the concise biomimetic synthesis, computational mechanistic insights, physicochemical attributes, kinetic parameters, molecular mechanism of action, and functional cellular targets of several phloroglucinol meroterpenoids. We harness synthetic clickable analogues of natural products to probe their disparate proteome-wide reactivity and subcellular localization through in-gel fluorescence scanning and cell imaging. By implementing sample multiplexing and a redesigned lysine-targeting probe, we streamline a quantitative activity-based protein profiling, enabling the direct mapping of global reactivity and ligandability of proteinaceous lysines in human cells. Leveraging this framework, we identify numerous lysine-meroterpenoid interactions in breast cancer cells at tractable protein sites across diverse structural and functional classes, including those historically deemed undruggable. We validate that phloroglucinol meroterpenoids perturb biochemical functions through stereoselective and site-specific modification of lysines in proteins vital for breast cancer metabolism, including lipid signaling, mitochondrial respiration, and glycolysis. These findings underscore the broad potential of phloroglucinol meroterpenoids for targeting functional lysines in the human proteome.
End of feed.